You don’t need to fear Chemistry anymore! We understand that Class 9 Chemistry can feel a bit tricky with new concepts like atoms, molecules, and the structure of matter. But worry not. We’ve brought you simplified notes that make every topic easy to understand. Download the latest CBSE Class 9 Science syllabus PDF for the current academic session
Class 9 Structure of Atom Notes
Class 9 builds the foundation for higher classes, so it’s important to have your basics clear from the start. These notes are designed to help you grasp concepts quickly, without any confusion. Whether it's exam time or a surprise test, you’ll be fully prepared.
Each topic is broken down in simple language, supported by key points and examples. Our goal is to help you learn better and score higher in your tests or exams with confidence.
Class 9 Structure of the Atom Notes
The chapter "Structure of the Atom" explains how atoms are made up of subatomic particles, electrons, protons, and neutrons.
It discusses the discovery of these particles and various atomic models like those of Thomson, Rutherford, and Bohr. Bohr’s model explains how electrons revolve in fixed energy levels.
The chapter also introduces concepts like atomic number, mass number, valency, isotopes, and isobars.
Discovery of Electron
J.J. Thomson discovered the electron in 1897 during his experiment with a cathode ray tube. When electricity was passed through gases at low pressure, a stream of particles moved from the negative electrode (cathode) to the positive one (anode).
These particles were negatively charged and were later named electrons. This discovery showed that atoms are not indivisible as once thought, but contain smaller particles. Electrons have a negative charge of -1.6 × 10⁻¹⁹ C and a very small mass compared to other subatomic particles.
Charge and Mass of Electron
Electrons are very light particles with a negligible mass of about 9.1 × 10⁻³¹ kg. They carry a negative charge, which is the smallest charge known. Their charge is considered as -1 unit, and they play a major role in chemical bonding and electricity.
Discovery of Proton and Neutron
This section explains how scientists discovered positively charged protons and neutral neutrons, completing our understanding of the internal structure of an atom.
Proton
Protons were discovered by E. Goldstein using canal rays, which are positively charged particles moving in the opposite direction of cathode rays. These rays revealed the presence of protons, which have a positive charge and a mass nearly 2000 times heavier than an electron.
- Charge on proton: +1
- Mass of proton: 1.67 × 10⁻²⁷ kg
Neutron
James Chadwick discovered neutrons in 1932. They are neutral particles (no charge) and they have mass slightly more than protons, and its mass is nearly equal to that of a proton.
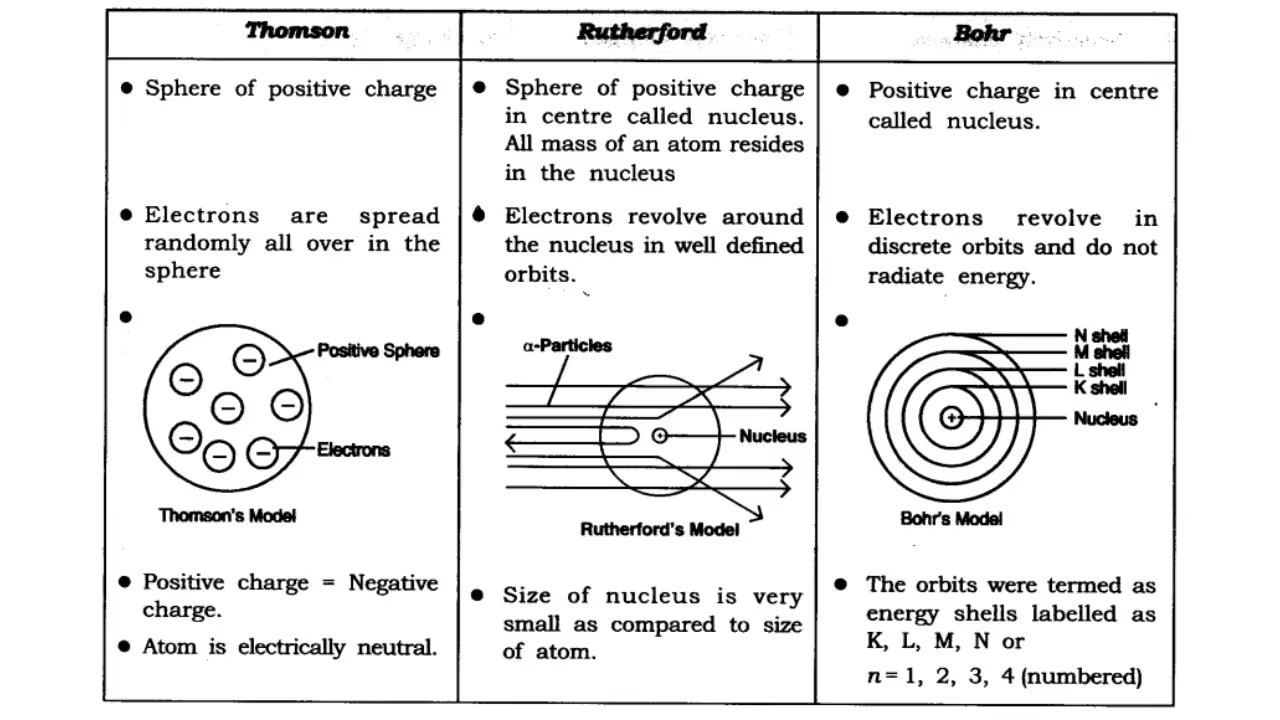
Thomson's Model of Atom
Thomson proposed that an atom is a positively charged sphere with electrons embedded inside it, just like seeds in a watermelon or plums in a pudding. This was known as the plum pudding model.
While it explained the presence of electrons and protons, it failed to explain how atoms remain stable and why some particles behave differently in experiments.
Rutherford’s Model of Atom
Ernest Rutherford conducted the famous alpha particle scattering experiment in which alpha particles were bombarded on a thin gold foil. Most passed straight through, but some were deflected and a few bounced back.
This led Rutherford to conclude that atoms have a small, dense, positively charged nucleus in the center, and electrons revolve around it. His model was like the solar system. However, it couldn't explain why the electrons don’t spiral into the nucleus due to attraction.
Drawbacks: Could not explain electron stability. Revolving electrons should lose energy and fall into the nucleus.
Bohr’s Model of Atom
To fix Rutherford’s model, Niels Bohr suggested that electrons revolve around the nucleus in fixed circular orbits or energy levels.
Main points:
- Electrons revolve in fixed orbits/shells around the nucleus.
- Each orbit has a fixed energy level.
- Electrons do not radiate energy in these fixed orbits.
- Energy is absorbed or emitted only when electrons jump between orbits.
This model successfully explained the stability of hydrogen atoms.
Distribution of Electrons in Different Shells (Bohr-Bury Rule)
Electrons are filled in shells in an atom using rule. 2n², where n = shell number gives the maximum number of electrons in a shell.
- K shell (n=1): 2 electrons
- L shell (n=2): 8 electrons
- M shell (n=3): 18 electrons
- Outer shell can have maximum 8 electrons
- Shells are filled step by step; one shell is filled before moving to the next.
Valency
Valency is the combining capacity of an atom. It depends on the number of electrons in the outermost shell.
- If outer shell has < 4 electrons: valency = number of valence electrons
- If outer shell has > 4 electrons: valency = 8 – number of valence electrons
Examples:
- Hydrogen (1 electron) → Valency = 1
- Oxygen (6 electrons) → Valency = 2
Atomic Number and Mass Number
The atomic number (Z) is the number of protons in an atom, which is also equal to the number of electrons in a neutral atom. The mass number (A) is the total number of protons and neutrons in the nucleus. So,
Mass number = Number of protons + Number of neutrons.
Atomic number helps in identifying the element, while mass number gives the total mass of the atom.
Don’t leave out important concepts and refer to the latest syllabus first.
Isotopes and Isobars
Isotopes are atoms of the same element with the same atomic number but different mass numbers. This happens because the number of neutrons is different. For example, hydrogen has three isotopes: Protium (1H¹), Deuterium (1H²), and Tritium (1H³).
Isobars, on the other hand, are atoms of different elements that have the same mass number but different atomic numbers. For example, argon (18Ar⁴⁰) and calcium (20Ca⁴⁰) are isobars.
FAQs
Q1. What was Thomson’s model of the atom?
Ans. Thomson said the atom looks like a plum pudding, with electrons scattered inside a positive sphere.
Q2. How did Bohr improve the atom model?
Ans. Niels Bohr said electrons revolve in fixed energy levels (or shells) without losing energy.
Q3. What did Rutherford’s model say?
Ans. He said atoms have a small, dense, positively charged nucleus at the center and electrons revolve around it.
Q4. What is the atomic number of an element?
Ans. Atomic number is the number of protons in an atom, which also equals the number of electrons.
Q5. What are valency and octet rule?
Ans. Valency is the combining capacity of an atom. The octet rule says atoms try to have 8 electrons in their outer shell to be stable.

.svg)





.avif)

.avif)
.avif)

.avif)











