Question:
Classify the following reactions into different types:
(i) AgNO3(aq) + NaCl(aq) AgCl(s) + NaNO3(aq)
(ii) CaO(s) + H2O(l) Ca(OH)2(aq)
(iii) 2KClO3(s) 2KCl(aq) + 3O2(g)
(iv) Zn + CuSO4 ZnSO4 + Cu
(B) Translate the following statement into a balanced chemical equation:
"Barium chloride reacts with aluminium sulphate to give aluminium chloride and barium sulphate."
Answer:
The balanced chemical equation for the reaction between barium chloride and aluminium sulphate to give aluminium chloride and barium sulphate is as follows:
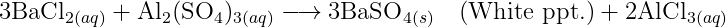

More NCERT Questions
Download Free PDF
(All Q's of this Chapter solved)
Related Topics

To Download PDF
Please verify your Whatsapp number first,
so you can download this pdf immediately
Please type a valid 10 digit whatsapp number

I am happy to receive premium PDFs
 Send OTP
Send OTPOTP sent, check your whatsapp
Your OTP is incorrect, Please enter valid OTP

.svg)










.avif)






