Ever felt confused about how invisible gas molecules can create something as real as pressure or temperature? Don’t worry - you’re not alone.
In Kinetic Theory of Gases Class 11 Notes, we break down this chapter in the simplest way possible. Instead of memorising formulas blindly, we’ll understand how tiny, constantly moving molecules explain gas laws, energy, and temperature.
This chapter from CBSE Syllabus Class 11 Physics is actually one of the most logical and scoring chapters in Physics - once the basics are clear, everything starts connecting beautifully.
Kinetic Theory of Gases Summary
Kinetic theory provides a microscopic picture of gases, describing them in terms of a very large number of tiny particles (molecules) that are in continuous random motion. This motion and the collisions of molecules explain macroscopic properties such as pressure, temperature, and volume.
Molecular Nature of Matter
All matter is composed of molecules, which are extremely small and separated by empty space. In gases, the intermolecular distances are very large compared to the size of the molecules, making gases highly compressible.
Evidence for the existence of molecules comes from Brownian motion, the zig-zag random motion of small suspended particles in fluids caused by bombardment from invisible fast-moving molecules. Another evidence is diffusion, the intermixing of two gases due to molecular motion.
Some important constants:
- Avogadro’s number (NA): 6.022×1023 particles per mole
- Universal gas constant (R): 8.314 J mol−1 K−1
- Boltzmann constant (kB): 1.38×10−23 J K−1
Gas Laws
Experiments on gases led to a set of empirical laws:
- Boyle’s Law: At constant temperature, p∝1/V.
- Charles’ Law: At constant pressure, V∝T.
- Gay-Lussac’s Law (Pressure Law): At constant volume, p∝T.
- Avogadro’s Law: Equal volumes of gases at the same temperature and pressure contain equal numbers of molecules.
Combining these laws gives the ideal gas equation:
pV = nRT = NkBT
where nn is the number of moles and N is the number of molecules.
Assumptions of Kinetic Theory
To model gases, kinetic theory assumes:
- Gas consists of identical point particles with negligible volume.
- Molecules are in random, continuous straight-line motion.
- Collisions between molecules and with container walls are perfectly elastic.
- Intermolecular forces are negligible except during collisions.
- The duration of a collision is negligible compared to the time between collisions.
- The number of molecules is very large, so averages give meaningful results.
Pressure of an Ideal Gas
According to kinetic theory, pressure arises due to continuous collisions of gas molecules with the walls of the container.
p = ⅓ ρ ⟨v2⟩
where ρ=Nm/v is the density of the gas, and ⟨v2⟩ is the mean square speed.
Kinetic Interpretation of Temperature
From kinetic theory:
½ m⟨v2⟩ = 3/2 kBT
This means:
Temperature is directly proportional to the average kinetic energy of molecules.
Higher temperature = faster molecular motion
U= 3/2 RT
So, the internal energy of an ideal monatomic gas depends only on temperature, not on pressure or volume.
Distribution of Molecular Speeds
Not all molecules move with the same speed. Maxwell–Boltzmann theory gives three characteristic speeds:
vp = √(2RT/M), vavg = √(8RT/πM) , vrms = √(3RT/M)
- vp: Most probable speed (maximum in distribution curve)
- vavg: Average speed
- vrms: Root mean square speed
They follow the relation: vp<vavg<vrms.
Law of Equipartition of Energy
According to this law, each quadratic degree of freedom contributes 12kBT\tfrac{1}{2}k_BT per molecule to the energy.
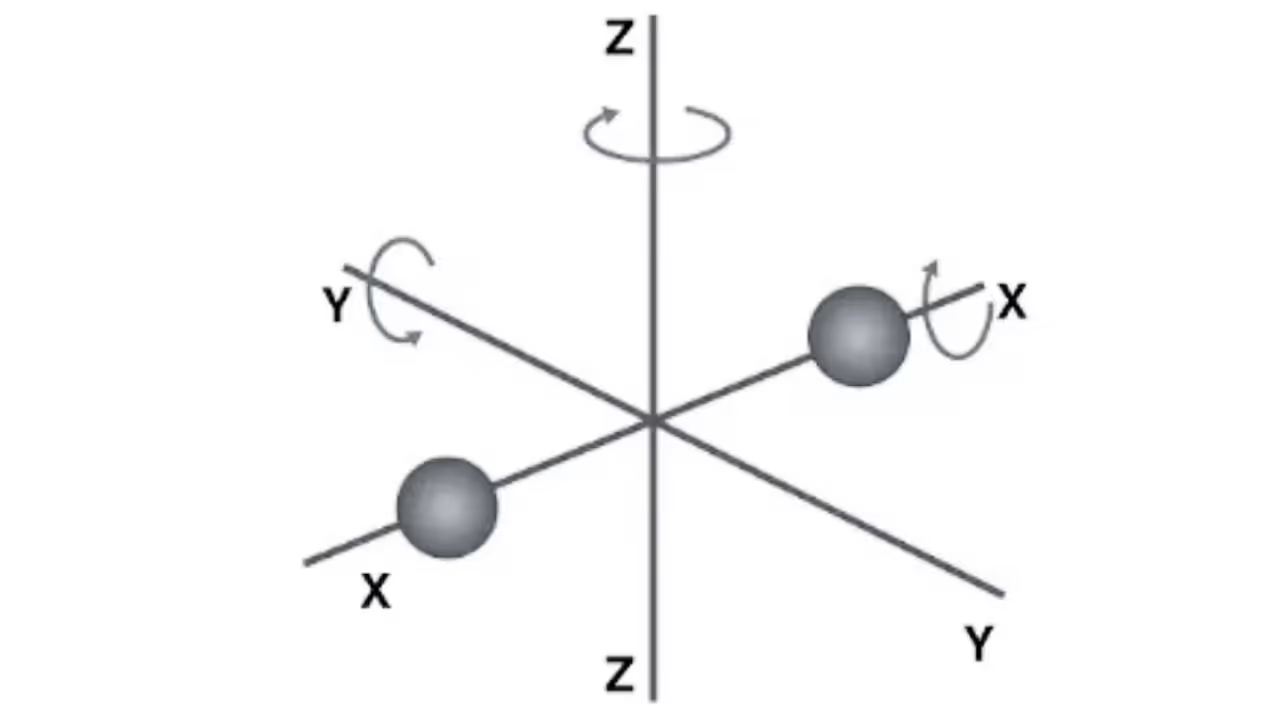
- Monatomic gas (He, Ne): 3 translational degrees → f = 3
U = 3/2nRT, Cv = 3/2R, Cp = 5/2R, γ = 5/3 - Diatomic gas (O₂, N₂) at room T: 3 translational + 2 rotational → f = 5
U = 5/2nRT, Cv = 5/2R, Cp = 7/2R, γ = 7/5 - Polyatomic non-linear gases: 3 translational + 3 rotational → f = 6
At high temperatures, vibrational modes activate and contribute additional energy.
Mean Free Path
The average distance a molecule travels between collisions is the mean free path λ.
λ = 1 / (√2 * π * d² * n)
where dd is molecular diameter and nn is number density.
At STP, λ is about 10−7 m.
Diffusion and Effusion
- Diffusion: Spreading of molecules from high concentration to low concentration due to random motion.
- Effusion: Escape of molecules through a tiny hole into vacuum.
Graham’s Law: Rate of diffusion/effusion is inversely proportional to the square root of molar mass.
Rate ∝ 1/√M
Real Gases and Deviations
Real gases deviate from ideal behaviour at:
- High pressure
- Low temperature
Reasons:
- Finite molecular volume
- Intermolecular attractive forces
Van der Waals corrected equation:
P + (an²/V²) (V - nb) = nRT
where a accounts for intermolecular attraction and b for finite volume.
Conclusion
That’s a wrap on Kinetic Theory of Gases. What looks like “just formulas” at first is actually a powerful idea - that the motion of tiny, invisible molecules explains everything about gases. From pressure to temperature, from RMS speed to internal energy, every concept is linked.
If you understand the logic behind molecular motion instead of mugging up equations, this chapter becomes one of the easiest scoring units in Class 11 Physics. Revise the key formulas once, understand the physical meaning behind them, and you’re exam-ready.
FAQs
Q1. What are the main assumptions of kinetic theory?
Ans. The assumptions include: gas molecules are point masses, they move randomly, collisions are perfectly elastic, intermolecular forces are negligible and pressure is due to collisions with container walls.
Q2. How is pressure explained in kinetic theory?
Ans. Pressure of a gas arises from the force exerted by molecules colliding with the walls of the container.
Q3. What are degrees of freedom in gases?
Ans. Degrees of freedom are the independent modes of motion (translational, rotational, vibrational) of a gas molecule in which it can store energy.
Q4. What is the law of equipartition of energy?
Ans. The law states that energy is equally distributed among all quadratic degrees of freedom, with each degree contributing ½ kBT per molecule.
Q5. How does kinetic theory explain specific heat of gases?
Ans. Specific heat depends on degrees of freedom. Monoatomic gases have only translational motion, while diatomic and polyatomic gases have translational, rotational and vibrational modes, giving different heat capacities.

.svg)










.avif)











