Thermodynamics is the branch of physics that deals with heat, work, temperature, and the laws governing energy transformation. Unlike mechanics, which studies motion, thermodynamics focuses on energy exchanges in macroscopic systems without considering microscopic molecular details.
This chapter from cbse syllabus class 11 physics explains how heat energy converts into mechanical work, the limitations of energy conversion, and the fundamental laws that govern these processes. It forms the foundation for understanding heat engines, refrigerators, entropy, and real-world energy systems.
Thermal Equilibrium and Zeroth Law of Thermodynamics
When two systems are placed in thermal contact, energy may flow between them in the form of heat until they attain the same temperature. At this stage, both are said to be in thermal equilibrium.
The Zeroth Law of Thermodynamics states that if two systems, A and B, are each in thermal equilibrium with a third system, C, then A and B are also in thermal equilibrium with each other.
This law provides the basis for the definition of temperature. It allows us to use a thermometer (the third system) to compare the temperatures of two different bodies.
Thermodynamic System
A thermodynamic system is a specified portion of the universe chosen for study. Types of systems:
- Open system: exchanges matter and energy
- Closed system: exchanges energy but not matter
- Isolated system: exchanges neither matter nor energy
Internal Energy (U)
Every system possesses energy due to the motion and configuration of its molecules. This is called the internal energy (U) of the system. It includes the kinetic energy of random molecular motion as well as the potential energy due to molecular interactions.
Internal energy is a state function, meaning it depends only on the current state of the system (like temperature, pressure, and volume), and not on the path by which that state was achieved.
Work
In thermodynamics, work is defined as the energy transferred by a system to its surroundings (or vice versa) when the external conditions such as pressure and volume change.
For example, consider a gas in a cylinder fitted with a piston. When the gas expands against an external pressure, the piston moves, and the gas does work on the piston. Conversely, if the piston compresses the gas, work is done on the gas.
The infinitesimal work done during an expansion or compression is given by ⇒ dW = ∫p dV; where p is the pressure of the system and dV is the change in volume.
Work depends on the path taken by the process, which means it is not a state function.
Heat
Heat is the form of energy transferred between two bodies at different temperatures due to the temperature difference alone. It flows from the body at higher temperature to the one at lower temperature until thermal equilibrium is established.
Like work, heat (Q) is also not a state function. The amount of heat absorbed or released depends on the path taken in the process.
First Law of Thermodynamics
The First Law of Thermodynamics is the law of conservation of energy applied to thermodynamic systems. It states that ⇒ ΔU = Q − W; where
- ΔU is the change in internal energy of the system,
- Q is the heat supplied to the system,
- W is the work done by the system on the surroundings.
This law implies that energy can neither be created nor destroyed; it can only be converted from one form to another. The total energy of an isolated system remains constant.
Specific Heat Capacity and Molar Heat Capacity
When heat is supplied to a system, its temperature rises. The amount of heat required to raise the temperature of a unit mass of the substance by one degree is called its specific heat capacity (c). Q = mc ΔT; where mm is mass, ΔT is the rise in temperature.
For gases, we often define molar heat capacities, which are the heat required to raise the temperature of one mole of gas by one degree. These are:
- Cp: Molar heat capacity at constant pressure
- Cv: Molar heat capacity at constant volume
For an ideal gas, they are related by the equation ⇒ Cp − Cv = R; where R is the universal gas constant.
Calorimetry
Water has a very high specific heat capacity, which is why it is used widely in cooling systems and temperature regulation in nature. The method of measuring heat exchange in physical and chemical processes is called calorimetry.
The principle of calorimetry is based on the conservation of energy: the heat lost by hot bodies equals the heat gained by cold bodies if there is no heat loss to the surroundings.
Isothermal and Adiabatic Processes
Thermodynamic processes can be classified based on how they take place:
- Isothermal Process: In this process, the temperature of the system remains constant (ΔT = 0), hence the internal energy does not change (ΔU = 0). The first law reduces to Q = W.
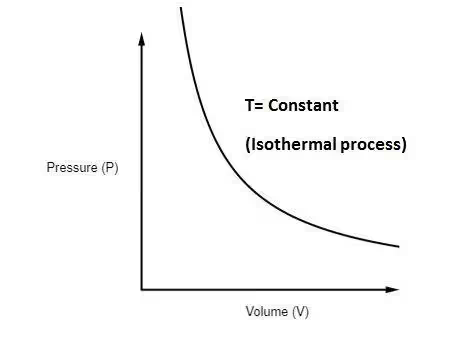
For an ideal gas undergoing isothermal expansion or compression ⇒ W = nRT ∫Vf/Vi
- Adiabatic Process: In this process, there is no heat exchange between the system and surroundings (Q=0). Therefore, ΔU = −W.
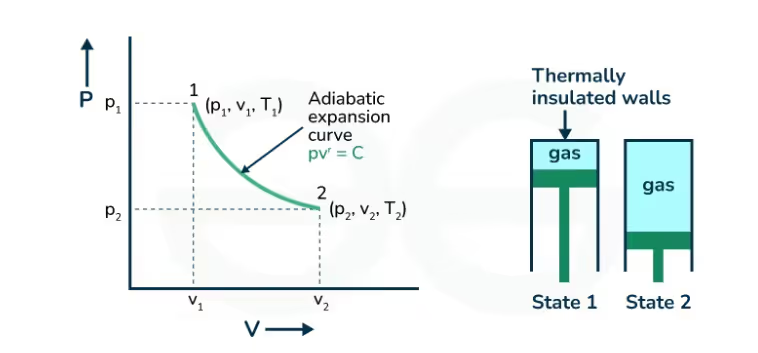
The relation between pressure and volume for an adiabatic process is ⇒ pVγ = constant; where γ = Cp/Cv is the adiabatic index.
Isobaric and Isochoric Processes
- Isobaric process is one that occurs at constant pressure. The heat supplied in such a process goes partly into doing work and partly into increasing the internal energy.
- Isochoric processes occur at constant volume (ΔV = 0), so no work is done (W = 0). The first law reduces to: Q = ΔU
The Second Law of Thermodynamics
While the first law tells us about energy conservation, it does not describe the direction of heat flow or the feasibility of processes. For this, we need the Second Law of Thermodynamics.
It can be stated in several equivalent forms:
- Kelvin-Planck Statement: It is impossible to construct a cyclic engine that extracts heat from a single reservoir and converts it entirely into work without any other effect.
- Clausius Statement: It is impossible to transfer heat from a colder body to a hotter body without the expenditure of external work.
The second law introduces the concept of irreversibility of natural processes.
Heat Engine
A heat engine is a device that converts heat energy into mechanical work in a cyclic process. It consists of:
- a hot reservoir at higher temperature,
- a working substance (like gas in a cylinder), and
- a cold reservoir at lower temperature.
The efficiency of a heat engine is defined as ⇒ η = W / QH = 1 − (Qc / QH); where QH is the heat absorbed from the hot reservoir and QC is the heat rejected to the cold reservoir.
Refrigerator and Heat Pump
A refrigerator is a device that operates on the reverse principle of a heat engine. It extracts heat from a cold reservoir and delivers it to a hot reservoir with the help of external work.
The performance of a refrigerator is measured by its Coefficient of Performance (COP) ⇒ COP = Qc / W; where QC is the heat extracted from the cold reservoir and WW is the work input.
Similarly, a heat pump transfers heat from a colder to a hotter body, but here the focus is on heating.
Carnot Engine
The Carnot Engine, proposed by Sadi Carnot, is an idealized heat engine that works on a reversible cyclic process consisting of two isothermal and two adiabatic processes. The cycle is called the Carnot cycle.
The efficiency of a Carnot engine depends only on the temperatures of the hot and cold reservoirs ⇒ η = 1 − TCTH, where TC and TH are the absolute temperatures of the cold and hot reservoirs.
This sets the maximum possible efficiency of any heat engine operating between two temperatures, and no real engine can surpass Carnot efficiency.
Entropy
Entropy is a measure of disorder or randomness of a system. The second law can also be expressed in terms of entropy ⇒ dS = dQ / T; for a reversible process.
In irreversible processes, the total entropy of the universe (system + surroundings) always increases. This is known as the principle of increase of entropy.
- For reversible process: ΔSuniverse = 0
- For irreversible process: ΔSuniverse > 0
Practical Applications of Thermodynamics
Thermodynamics has numerous practical applications:
- Heat engines in automobiles and power plants
- Refrigerators and air conditioners
- Thermal power stations
- Industrial heat transfer systems
- Natural processes like weather cycles
Conclusion
Thermodynamics establishes the fundamental laws governing energy transformation. The Zeroth Law defines temperature, the First Law ensures energy conservation, and the Second Law determines the direction and limitations of energy conversion.
Concepts such as entropy, heat engines, and Carnot efficiency provide deep insight into the efficiency of natural and artificial systems. This chapter forms the conceptual foundation for advanced thermodynamics and modern physics applications.
FAQs
Q1. What is the First Law of Thermodynamics?
Ans. ΔU = Q − W. It expresses conservation of energy in thermodynamic systems.
Q2. What is the Second Law of Thermodynamics?
Ans. It states that heat cannot be completely converted into work in a cyclic process and introduces entropy.
Q3. What is the Third Law of Thermodynamics?
Ans. The third law states that the entropy of a pure substance approaches zero as its temperature approaches absolute zero.
Q4. What is an isothermal process?
Ans. An isothermal process occurs at constant temperature, where internal energy remains constant and heat supplied is fully converted into work.
Q5. What is an adiabatic process?
Ans. An adiabatic process is one in which no heat is exchanged with the surroundings, so the change in internal energy equals the work done.

.svg)










.avif)











