Every time a chemical reaction takes place, whether it's your food getting digested or fuel burning in a car, energy is either absorbed or released. But how do we measure this energy? Can we tell if a reaction will happen on its own? These are some of the big questions this chapter will answer.
In simple terms, thermodynamics is the study of energy changes in chemical and physical processes. It doesn’t look at how fast a reaction happens or how the atoms move, but only at the beginning and the end, what goes in and what comes out. Get complete overview from CBSE Class 11 Chemistry Syllabus.
By the end of this chapter, you’ll have a better idea of how energy drives changes in nature, in your body, and even in the machines around you. It’s like learning the rules behind every chemical change that makes life possible!
Introduction to Thermodynamics
Thermodynamics is the branch of science that deals with the study of energy changes, particularly the transformation of heat into other forms of energy and vice versa.
It helps us understand how and why chemical reactions occur and allows us to predict whether a reaction will happen under given conditions.
Thermodynamics does not consider the rate at which a reaction occurs or the molecular-level mechanisms of reactions. It is a macroscopic science, meaning it deals with the overall system rather than individual atoms or molecules.
Zeroth Law of Thermodynamics
If two systems are separately in thermal equilibrium with a third system, then they are in thermal equilibrium with each other. This law helps define the concept of temperature.
System and Surroundings
A system refers to the part of the universe in which observations are made. Everything else apart from the system is considered the surroundings.
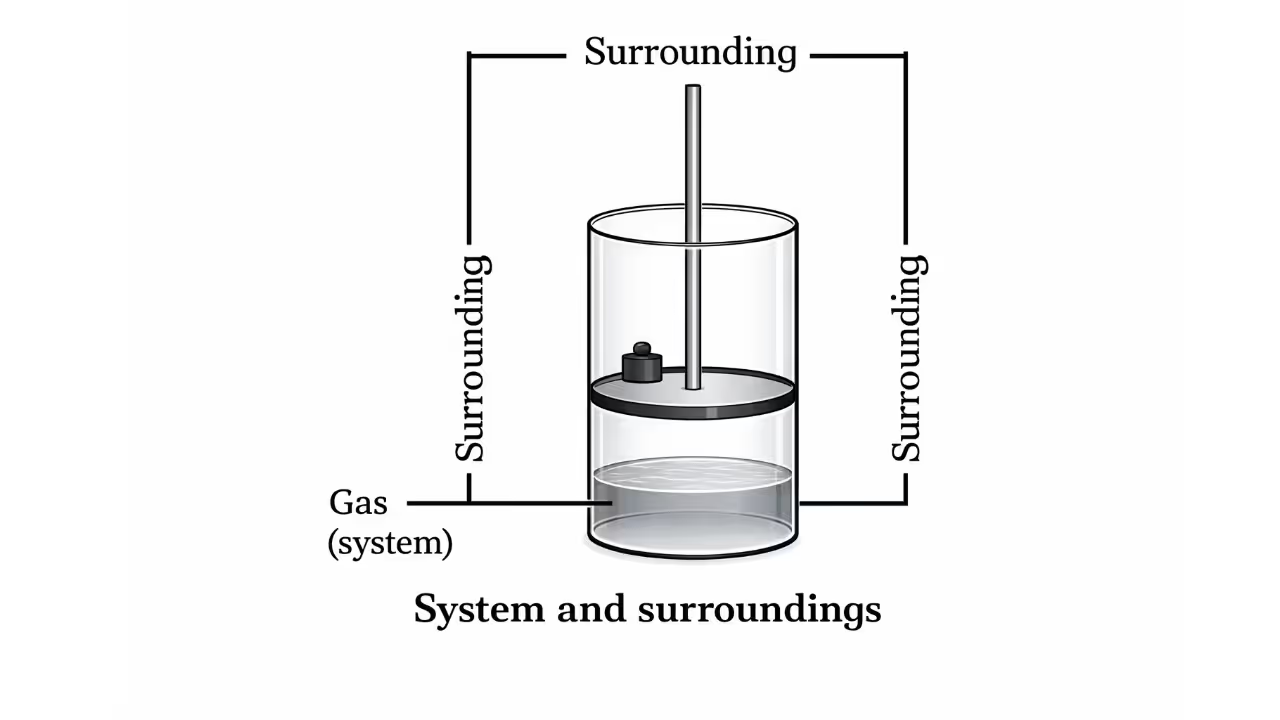
Systems are classified into three types:
- Open System - Both energy and matter can be exchanged with the surroundings. Example: A boiling pot of water without a lid.
- Closed System - Only energy (not matter) can be exchanged with the surroundings. Example: Water boiling in a closed container.
- Isolated System - Neither energy nor matter is exchanged with the surroundings. Example: A thermos flask.
The boundary between the system and the surroundings can be real or imaginary and can be fixed or movable.
Types of Properties
Properties of a system are measurable quantities used to describe its state.
- State functions are properties that depend only on the current state of the system and not on how that state was achieved. Examples include pressure (P), volume (V), temperature (T), internal energy (U), enthalpy (H), and entropy (S).
- Path functions depend on the path or route taken to reach a particular state. Heat (q) and work (w) are path functions.
Extensive and Intensive Properties
- Extensive properties depend on the amount of substance present.
Examples: mass, volume, internal energy. - Intensive properties do not depend on the amount of substance.
Examples: temperature, pressure, density.
Internal Energy (U)
Every system possesses a certain amount of energy, called internal energy. It includes the kinetic and potential energies of all the particles in the system. Internal energy can be changed by:
- Heat exchange (q)
- Work done (w)
The first law of thermodynamics expresses this as:
ΔU = q + w
For ideal gases, internal energy depends only on temperature.
ΔU = nCvΔT
Where:
- ΔU is the change in internal energy,
- q is heat absorbed by the system (+ve if heat is absorbed),
- w is the work done on the system (+ve if work is done on the system).
This law is a statement of conservation of energy: energy can neither be created nor destroyed, only transformed.
Work Done in Thermodynamics
Work is said to be done when there is a movement against an opposing force. In thermodynamics, we mainly consider pressure-volume work.
For an infinitesimal change:
dw = –Pext × dV
For finite change at constant external pressure:
w = -Pext (V2 - V1)
If gas expands → work is negative (system does work).
If gas is compressed → work is positive (work done on system).
Important: For reversible expansion of ideal gas:
w = –nRT ln(V₂/V₁)
Heat (q)
Heat is energy transferred between a system and its surroundings due to a temperature difference. It flows from a higher to a lower temperature region. Like work, it is also a path function.
The amount of heat absorbed or released at constant pressure or volume differs:
- At constant volume: qᵥ = ΔU
- At constant pressure: qₚ = ΔH
Enthalpy (H)
Enthalpy is a thermodynamic quantity equivalent to the total heat content of a system. It is defined as:
H = U + PV
Change in enthalpy:
ΔH = ΔU + PΔV
At constant pressure, the heat exchanged equals the change in enthalpy:
qₚ = ΔH
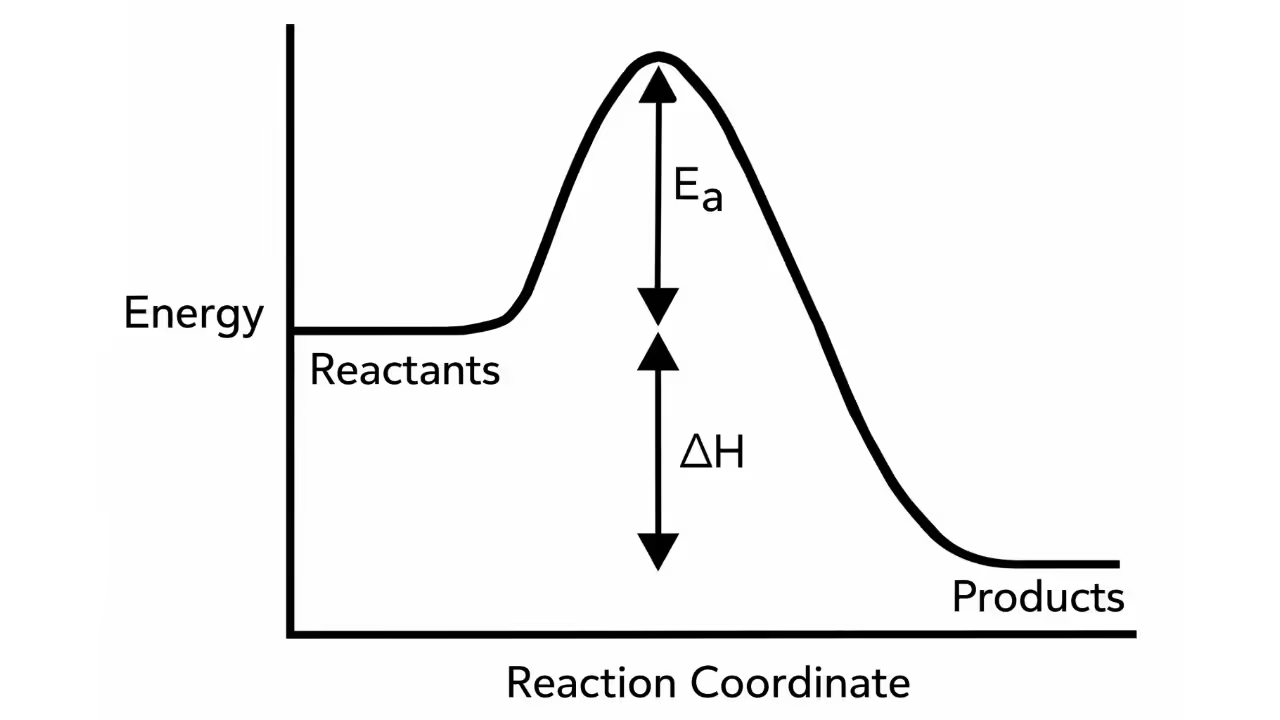
- ΔH > 0 → Endothermic process (absorbs heat)
- ΔH < 0 → Exothermic process (releases heat)
Heat Capacity and Specific Heat
Heat capacity (C) is the amount of heat required to raise the temperature of a system by 1 K (or 1°C).
C = q / ΔT
- At constant volume: qv = nCvΔT
- At constant pressure: qp = nCpΔT
For an ideal gas:
Cₚ – Cv = R, where R is the universal gas constant.
Measurement of ΔU and ΔH – Calorimetry
Calorimetry is an experimental technique to measure the amount of heat exchanged in a chemical or physical process.
The device used is called a calorimeter.
- At constant volume (bomb calorimeter): qᵥ = ΔU
- At constant pressure (open vessel): qₚ = ΔH
Hess’s Law of Constant Heat Summation
According to Hess's Law, the total enthalpy change for a reaction is the same whether it occurs in one step or in several steps.
If reaction A → D can occur via multiple steps:
- A → B → C → D
Then: ΔH(total) = ΔH1 + ΔH2 + ΔH3
This law is useful in calculating enthalpy changes for reactions that are difficult to perform directly.
Enthalpies of Common Reactions
- Enthalpy of formation (ΔH°f): Heat changes when one mole of a compound is formed from its elements in their standard states.
- Enthalpy of combustion (ΔHc°): Heat released when one mole of a substance burns completely in oxygen.
- Enthalpy of neutralization: Heat change when one mole of H⁺ reacts with one mole of OH⁻ to form water.
- Enthalpy of atomization: Heat required to break one mole of a substance into its atoms in the gaseous state.
Spontaneity of a Process
A process is spontaneous if it occurs on its own without needing continuous external energy.
Spontaneity depends on two factors:
- Enthalpy change (ΔH) – Most spontaneous reactions are exothermic (ΔH < 0).
- Entropy change (ΔS) – Spontaneity also depends on the degree of disorder or randomness.
A reaction is spontaneous when:
ΔG = ΔH – TΔS < 0
Cases:
- ΔH negative, ΔS positive → always spontaneous
- ΔH positive, ΔS negative → never spontaneous
- Both positive or both negative → depends on temperature
Even endothermic reactions can be spontaneous if there is a sufficient increase in entropy.
Entropy (S)
Entropy is a measure of randomness or disorder in a system.
- Greater disorder → higher entropy.
- In any natural process, total entropy tends to increase.
Change in entropy is given by:
ΔS = qrev / T (for reversible processes)
Entropy is a state function.
Gibbs Free Energy (G)
To predict spontaneity, Gibbs free energy is used:
G = H – TS
Change in free energy:
ΔG = ΔH – TΔS
- If ΔG < 0 → process is spontaneous
- If ΔG = 0 → process is in equilibrium
- If ΔG > 0 → process is non-spontaneous
Standard Gibbs Free Energy Change (ΔG°)
The standard free energy change for a reaction is:
ΔG° = ΔH° – TΔS°
This is used to determine whether a reaction is spontaneous under standard conditions (1 atm pressure, 298 K temperature).
Conclusion
That’s a wrap on Chemical Thermodynamics. This chapter teaches you how energy changes control every chemical reaction - whether it will release heat, absorb heat, or even occur at all. From internal energy to Gibbs free energy, every concept connects like pieces of a puzzle.
If you understand ΔU, ΔH, ΔS and ΔG clearly, most board numericals become straightforward. Revise the formulas, understand the sign conventions properly, and you’re already ahead. I hope this made the chapter feel less scary and more logical. Keep revising - you’ve got this.
FAQs
Q1. What is chemical thermodynamics?
Ans. Chemical thermodynamics is the study of energy changes during chemical reactions and physical processes. It helps us understand heat exchange and predict whether a reaction will occur.
Q2. What is the first law of thermodynamics?
Ans. The first law states that energy can neither be created nor destroyed. It only changes from one form to another, expressed as ΔU = q + w.
Q3. What is the difference between ΔH and ΔU?
Ans. ΔU is the change in internal energy of a system. ΔH is the heat change at constant pressure.
Q4. What makes a reaction spontaneous?
Ans. A reaction is spontaneous if it occurs on its own without continuous external energy. It happens when Gibbs free energy (ΔG) is negative.
Q5. What is the formula of Gibbs free energy?
Ans. The formula is ΔG = ΔH – TΔS. If ΔG is negative, the process is spontaneous.

.svg)










.avif)











