Chemical Bonding and Molecular Structure explains why atoms combine and how molecules are formed. Except for noble gases, most atoms are unstable in their isolated state and tend to gain, lose, or share electrons to achieve stability.
This chapter from Chemistry Syllabus Class 11 builds the foundation for understanding molecular geometry, bond strength, bond order, hybridisation, and intermolecular forces.
Concepts like VSEPR theory, Valence Bond Theory (VBT), and Molecular Orbital Theory (MOT) help explain not just bond formation but also molecular shape and stability, making this chapter highly important for both CBSE board exams and competitive exams like JEE and NEET.
Kössel and Lewis Approach to Chemical Bonding
In 1916, Kössel and Lewis provided the first modern explanation of chemical bonding, rooted in the newly discovered electronic structure of atoms.
Kossel observed that during the formation of certain compounds, atoms tend to lose or gain electrons to acquire the stable configuration of the nearest noble gas.
For example:
Na → Na⁺ + e⁻
Cl + e⁻ → Cl⁻
Na⁺ + Cl⁻ → NaCl
Electrostatic attraction between oppositely charged ions forms an ionic bond.
Lewis, on the other hand, focused on covalent bonds, where atoms share pairs of electrons to attain noble gas configurations. He introduced the Lewis dot structure, in which valence electrons are represented as dots around the chemical symbol of the element.
Octet Rule:
Both Kossel and Lewis emphasized that atoms tend to achieve eight electrons in their outer shell (the octet) for stability, similar to noble gases.
Limitations of Octet Rule:
- Incomplete octet → BeCl2, BF3
- Expanded octet → PCl5, SF6
- Odd-electron molecules → NO, NO2
- Does not explain molecular shapes
Types of Chemical Bonding
Chemical bonds are forces holding atoms together to form molecules or compounds, with the main types being ionic, covalent, and metallic.
1. Ionic (Electrovalent) Bond
An ionic bond is formed through the complete transfer of electrons from one atom to another, resulting in the formation of positively and negatively charged ions. Electrostatic forces of attraction hold these ions together in a crystalline lattice.
Characteristics:
- Usually occurs between metals and non-metals
- Large electronegativity difference between the atoms (generally > 1.7 on Pauling’s scale)
- Strong and directional only in the lattice form
Example: NaCl → Sodium loses one electron, chlorine gains one electron, forming Na⁺ and Cl⁻ ions.
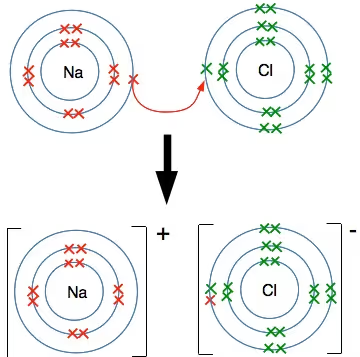
Factors affecting the formation of ionic bonds:
- Ionization Enthalpy: Lower ionization enthalpy of the metal favors bond formation
- Electron Gain Enthalpy: Higher electron affinity of the non-metal increases the tendency to form anions
- Lattice Enthalpy: High lattice energy stabilizes the ionic solid
2. Covalent Bond
A covalent bond forms when two atoms share one or more pairs of electrons to achieve noble gas configurations.
Characteristics:
- Usually formed between non-metal atoms
- Directional in nature (due to orbital overlap)
- Electrons are localized between the bonded atoms
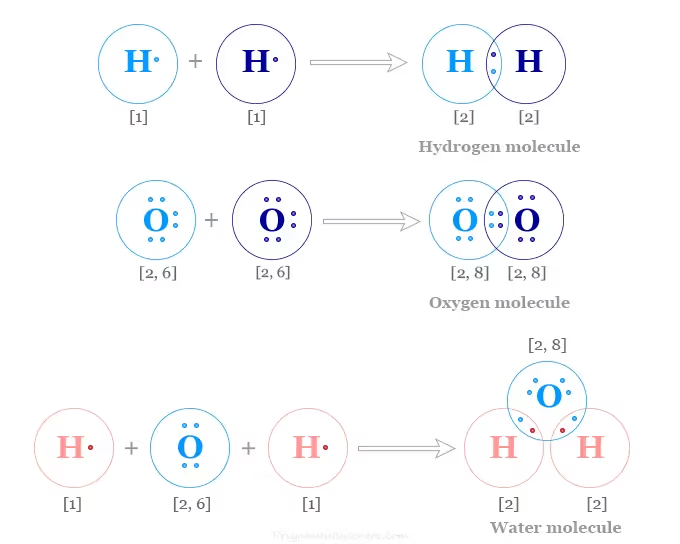
Single, Double, and Triple Bonds:
Depending on the number of electron pairs shared, bonds are classified as:
- Single bond – one pair shared (H₂, Cl₂)
- Double bond – two pairs shared (O₂)
- Triple bond – three pairs shared (N₂)
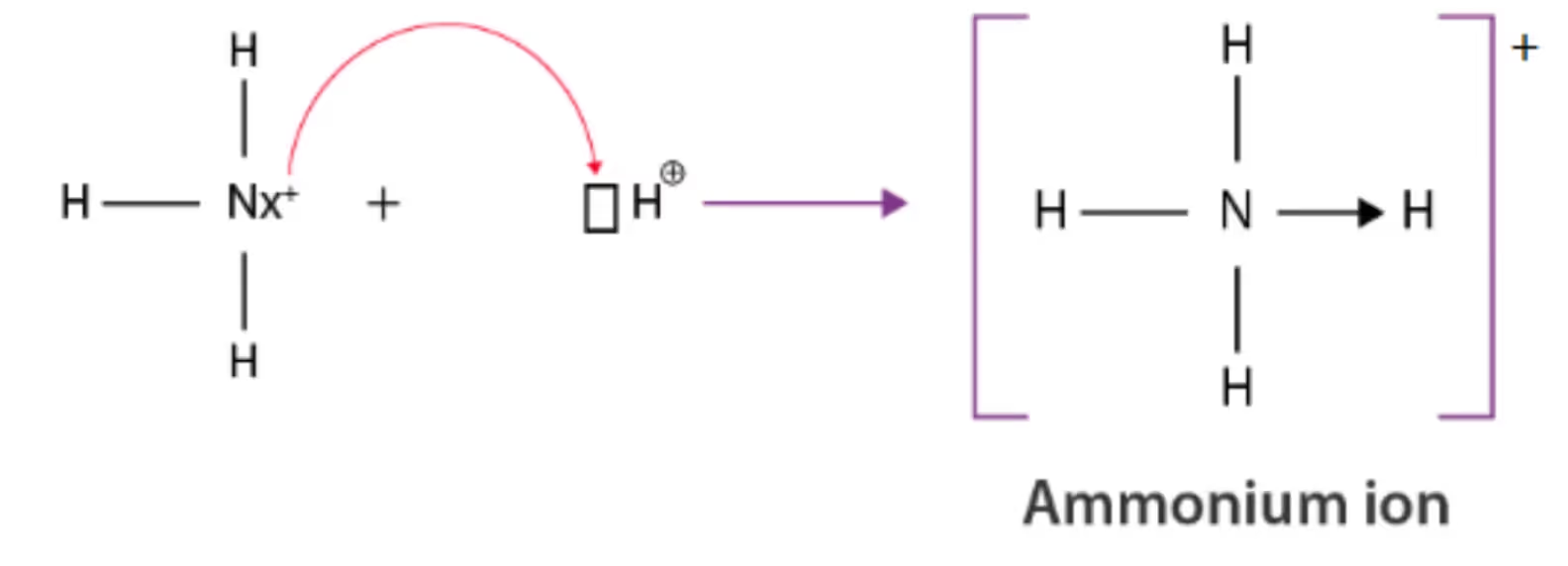
3. Coordinate (Dative) Bond
A coordinate bond is a type of covalent bond in which both shared electrons come from the same atom. Once formed, it is indistinguishable from a covalent bond.
Example: Formation of NH₄⁺ from NH₃ and H⁺.
4. Metallic Bond
Proposed by the electron sea model, metallic bonds arise from the attraction between positively charged metal ions and a ‘sea’ of delocalized electrons. This explains properties like:
- Malleability of metals
- Ductility of metals
- Electrical conductivity of metals
![image3.png [apng-to-avif output image]](https://cdn.prod.website-files.com/5f5cf4627107791c0412287b/699961ce368e69fcd7b43c24_ezgif-340865b1040a2c84.avif)
Bond Parameters
This was missing depth earlier. Add this properly.
1. Bond Length
Distance between nuclei of two bonded atoms–increases with atomic size and decreases with bond order.
Triple bond < Double bond < Single bond
2. Bond Angle
Angle between two bonds around a central atom. For example:
- CH₄ → 109.5°
- NH₃ → 107°
- H₂O → 104.5°
3. Bond Enthalpy
Energy required to break a bond in gaseous state. Higher bond enthalpy → stronger bond
4. Bond Order
Bond Order = (Nb − Na) / 2; where:
- Nb = number of electrons in bonding molecular orbitals
- Na = number of electrons in antibonding molecular orbitals
Higher bond order → greater stability
Valence Shell Electron Pair Repulsion (VSEPR) Theory
Proposed by Sidgwick and Powell, VSEPR theory predicts the shape of molecules based on the repulsion between electron pairs (bonding and lone pairs) in the valence shell.
Basic idea: Electron pairs repel each other and arrange themselves as far apart as possible to minimize repulsion.
Order of repulsion strength:
Lone pair–lone pair > lone pair–bond pair > bond pair–bond pair
Examples of molecular shapes:
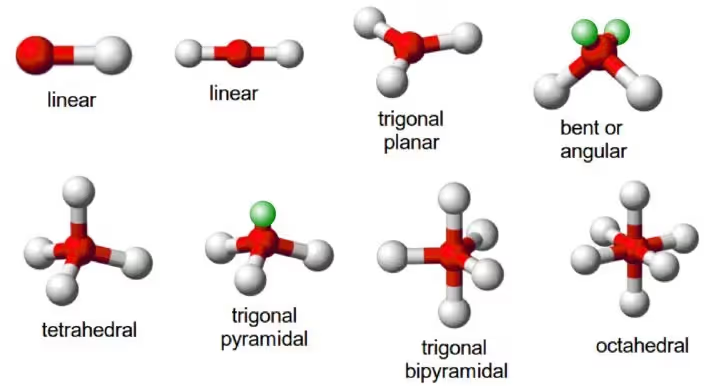
Common Shapes:
Valence Bond Theory (VBT)
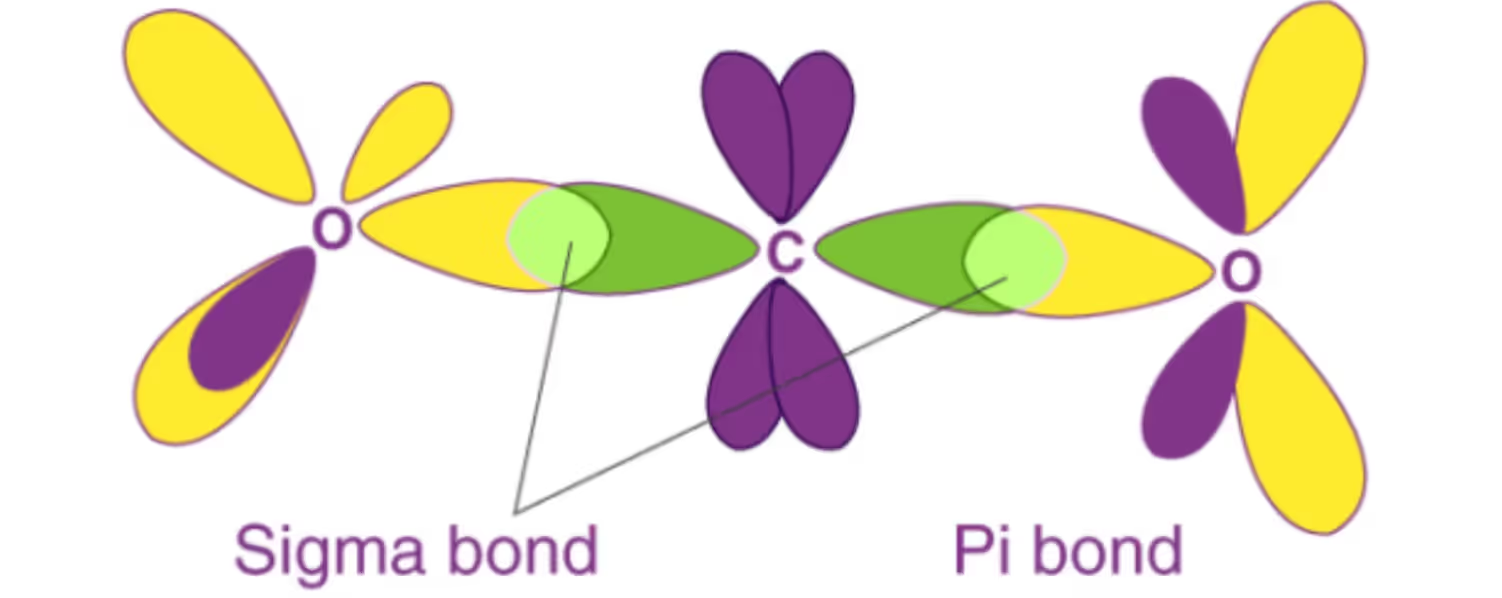
Proposed by Heitler and London and later refined by Pauling, VBT explains covalent bond formation as a result of overlap of half-filled atomic orbitals.
Types of overlaps:
- Sigma (σ) bond: Formed by head-on (axial) overlap; stronger bond.
- Pi (π) bond: Formed by sideways overlap of p-orbitals; weaker than σ bonds.
Hybridization: Mixing of atomic orbitals to form new hybrid orbitals of equivalent energy.
Molecular Orbital Theory (MOT)
Molecular Orbital Theory (MOT), developed by Hund and Mulliken, explains bonding in terms of molecular orbitals formed by the linear combination of atomic orbitals (LCAO).
Key points:
- Bonding MO has lower energy than atomic orbitals, anti-bonding MO has higher energy.
- Bond order = (Number of electrons in bonding MO − Number in antibonding MO)/2
- Bond order indicates bond strength and stability.
- If Bond Order = 0 → molecule does not exist.
Hydrogen Bonding
Hydrogen bonding is a special type of dipole–dipole attraction between hydrogen attached to a highly electronegative atom (F, O, or N) and the lone pair of another electronegative atom.
Types:
- Intermolecular: Between molecules (e.g., H₂O molecules).
- Intramolecular: Within the same molecule (e.g., o-nitrophenol).
Responsible for:
- High boiling point of water
- Structure of DNA
- Protein folding
Conclusion
Chemical bonding explains the fundamental forces that hold matter together. By understanding ionic, covalent, metallic bonding, and advanced theories like VSEPR, VBT, and MOT, students can predict molecular geometry, stability, and reactivity.
This chapter forms the conceptual backbone for higher studies in organic, inorganic, and physical chemistry.
FAQs
Q1. What is a covalent bond?
Ans. A covalent bond is formed when two atoms share one or more pairs of electrons to attain stability. Example: H2O, CH4.
Q2. What is the difference between ionic and covalent bonds?
Ans. Ionic bond involves transfer of electrons, while covalent bond involves sharing of electrons.
Q3. What is the VSEPR theory?
Ans. The Valence Shell Electron Pair Repulsion (VSEPR) theory predicts the shape of molecules based on the repulsion between electron pairs around the central atom.
Q4. What is the difference between sigma (σ) and pi (π) bonds?
Ans. A sigma bond is formed by head-on overlap of orbitals and is stronger, while a pi bond is formed by sideways overlap of orbitals and is weaker.
Q5. What is molecular orbital (MO) theory?
Ans. MO theory explains bonding by combining atomic orbitals to form molecular orbitals, which can be bonding, antibonding or non-bonding.

.svg)










.avif)











