Question:
What effect does the concentration of H+ (aq) ions have on the nature of the solution?
Answer:
Concentration of H+ (hydrogen) ions
- To know the nature of the solution, i.e., basic or acidic, the concentration of ions is needed.
- To identify the solution, a pH value or the concentration of H+ ions is used.
- The concentration of H+ ions increases as the solution turns acidic, whereas the concentration of H+ ions decreases when the solution is basic/alkaline.
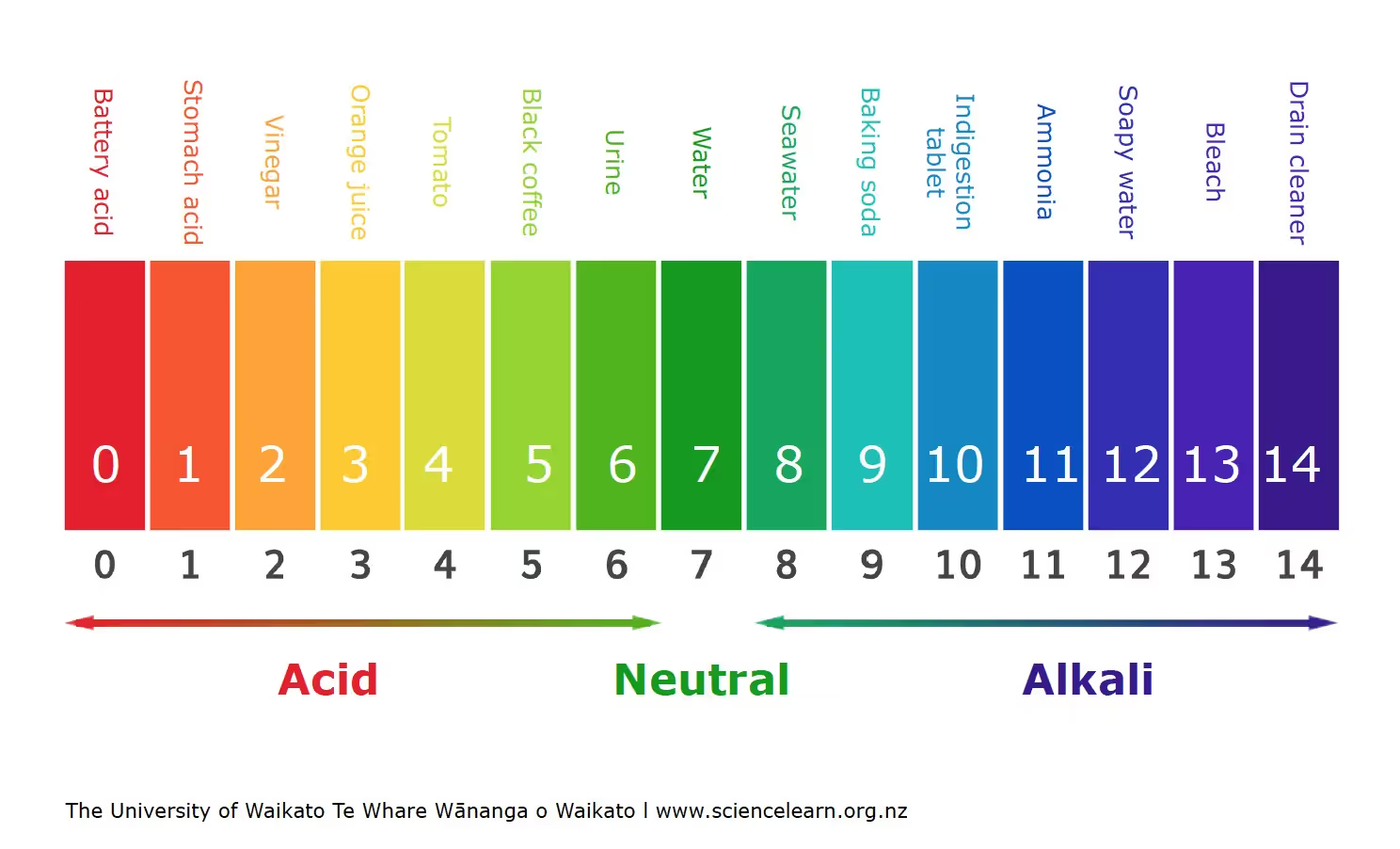
pH Scale
- It determines the measure of the hydrogen ion H+ concentration.
- The solution is acidic if the value is less than 7, neutral when it's 7, and if it's greater than 7, the solution is basic in nature.

.svg)





.avif)

.avif)
.avif)

.avif)









