Question:
Write the electronic configuration of any one pair of isotopes and isobar
Answer:
Isotopes are atoms having the same atomic number but have different mass numbers. So their electronic configuration will be the same. For example carbon has two isotopes 12 6C 13 6C , as the configuration requires only no of electrons and isotopes have the same number of electrons.
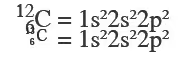
Isobars are atoms of different elements having the same mass number i.e. protons and neutrons. For example Argon and Calcium are the Isobar of each other. As the configuration requires only no of electrons and Isobars have a different number of electrons so their configuration varies.
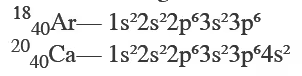

.svg)





.avif)

.avif)
.avif)

.avif)









