Question:
When 3.0g of carbon is burnt in 8.00g oxygen, 11.00g of carbon dioxide is produced what mass of carbon dioxide will be formed when 3.00g of carbon is burnt in 50.00g of oxygen ? Which low of chemical combination will govern your answer ?
Answer:
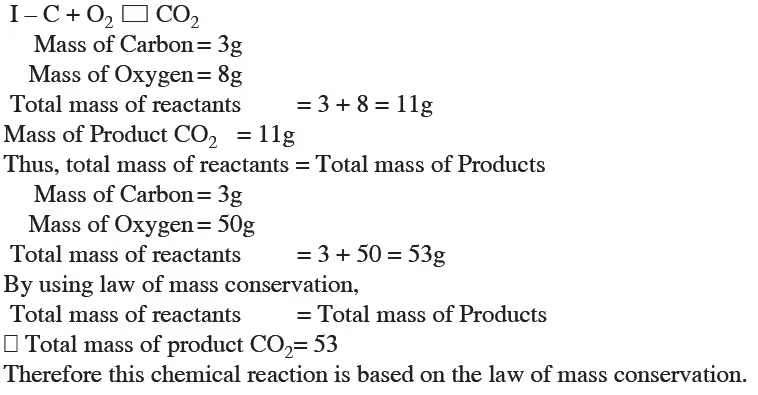

More NCERT Questions
Download Free PDF
(All Q's of this Chapter solved)
Related Topics

To Download PDF
Please verify your Whatsapp number first,
so you can download this pdf immediately
Please type a valid 10 digit whatsapp number

I am happy to receive premium PDFs
 Send OTP
Send OTPOTP sent, check your whatsapp
Your OTP is incorrect, Please enter valid OTP

.svg)





.avif)

.avif)
.avif)

.avif)





