Question:
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Answer:
Covalent bond formation in CH3Cl
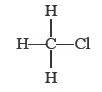
In this structure three hydrogen atoms are linked to carbon by covalent bond and there is also covalent bond between carbon and chlorine, but chlorine is more negative than carbon so it forms a polar covalent bond.
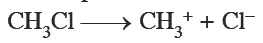

More NCERT Questions
Download Free PDF
(All Q's of this Chapter solved)
Related Topics

To Download PDF
Please verify your Whatsapp number first,
so you can download this pdf immediately
Please type a valid 10 digit whatsapp number

I am happy to receive premium PDFs
 Send OTP
Send OTPOTP sent, check your whatsapp
Your OTP is incorrect, Please enter valid OTP

.svg)





.avif)

.avif)
.avif)

.avif)





